
Be a part of the conversation and get involved.
You have 0 item in your basketTotal £0.00
You have no items in your basket
Be part of the solution and make a difference
Organisation or institution: International Institute for Active Ageing
Contact details: [email protected]
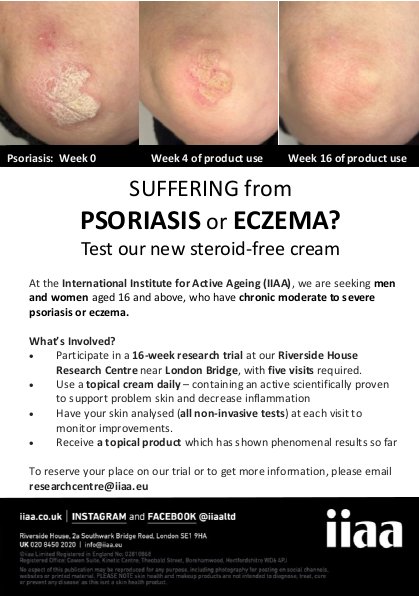
Description of activity: We are a company that conducts research and provides services for skin health. We are conducting a paid research trial for a steroid-free product that has been shown to reduce psoriasis.
What’s Involved?
Recruit criteria: Participants aged 16 and above with moderate to severe psoriasis.
Where? London Bridge.
Time commitment: 16 weeks, five in-person appointments.
Closing date: Ongoing study. (We want to recruit 15 individuals with psoriasis.)
Other information: We would require images of the affected areas of the participants so we can assess their severity.
Funder/sponsor: International Institute for Active Ageing.

Please note:
Organisation or institution: University of the West of England
Main study contact: Christine Silverthorne
Public contact details: [email protected]
Description of activity: We want to hear your views about what self-management means to you, the support you’ve been offered, and helpful this was to you in the first few years of being diagnosed. We want to understand the challenges faced by people newly diagnosed with psoriatic arthritis and how helpful self-management support is in meeting these challenges. We also want to discuss whether you feel this support could be improved and if so, your thoughts and ideas on how. Each discussion group will consist of 4 to 6 people living with psoriatic arthritis and will last for between 60 to 90 minutes. Participants will receive a £25 voucher as reimbursement for their time. What you tell us will help us plan future work around meeting the support needs of people newly diagnosed with psoriatic arthritis. These discussion groups are part of the TULiPs Study
Recruit criteria: We are looking for patients recently diagnosed with psoriatic arthritis to take part in an online discussion group and help us with our research about self-management support for people with psoriatic arthritis.
Where? Online
Time commitment: 60 to 90 minutes
Closing date: 30th June 2025
Funder/sponsor: National Institute for Health Research NIHR


Please note:


Please note:
Organisation or institution: Otoimmune & Thiscovery
Main study contact: Jenni Burt
Public contact details: [email protected]
Description of activity: Otoimmune was set up by individuals who either live with autoimmune conditions themselves or have close family members who do. They are seeking to develop tools and resources to help everyone impacted to better understand, manage, and improve their well-being. To help in this aim, they are gathering insights from people with these conditions, with symptoms, their family members and carers, and anyone proactively managing their health. They want to hear what experiences have been, what might better support, and about interests in a technological solution.
Recruit criteria: Someone who has been diagnosed with an autoimmune condition. Someone who suspects they may have an autoimmune condition. Family members of someone with an autoimmune condition Health-conscious individuals.
Where? Online via survey link
Time commitment:15 minutes
Closing date: 28/3/25
Funder/sponsor: Otoimmune
Please note:
Organisation or institution: Trinity College Dublin, Ireland
Main study contact: Olivia Headon
Public contact details: [email protected]
Description of activity: We are conducting a study to understand how immune-related conditions like eczema (atopic dermatitis), psoriasis and Crohn’s disease might impact mental health, specifically looking at symptoms of depression and obsessive-compulsive disorder (OCD).
This study aims to contribute to the research that is available to healthcare providers as they develop more targeted support for people living with these conditions. Participants will complete an anonymous online survey including questions about their condition, well-being and experiences.
This survey should take around 15 minutes. Responses are entirely anonymous, and no personal identifying information will be shared with the researcher.
Any questions: You can reach out to [email protected] with any questions related to this study. Withdrawal: You can withdraw from the study by simply not finishing the survey and exiting from the webpage.
Data Use: All data collected is anonymous and will be used only for research purposes. Your answers could contribute to a better understanding of the mental health challenges faced by people living with three common immune-mediated inflammatory conditions. Findings from this study could help shape future support and treatments by contributing to the information available to practitioners and other researchers.
Recruit criteria: English speakers living anywhere in the world between the ages of 18-65 and who either: Have a diagnosis of eczema, psoriasis or Crohn’s disease Do not have an immune-mediated inflammatory disease to serve as a comparison.
Where? Online survey (remote).
Time commitment: No more than 15 minutes.
Closing date: 17 February 2025.
Funder/sponsor: None.

Please note:
Organisation or institution: London South Bank University, (psychology department)
Main study contact: Reyhan Turan
Public contact details: [email protected]
Description of activity: Participants will be invited to take part in a 45-minute online interview. To ensure ethical standards, consent forms will need to be signed, granting permission to use the data for research purposes. The interview is completely voluntary and free of charge, with no compensation provided. This research is part of my undergraduate degree in Clinical Psychology. If needed, I will offer support to participants at the end of the interview. The study has received full ethical approval, and I can provide proof of this if requested.
Recruit criteria: Participants over 18, living with chronic pain for more than 6 months, and fluent in English,
Where? Remote, Teams or Zoom
Time commitment: 45-60 minutes max
Closing date: 1st February 2025
Funder/sponsor: None.


Please note:
Organisation or institution: Skin of Colour Training (SOCT) UK
Contact details: Dr Marisa Taylor & Dr Dami Jagun Co-Founders of Skin of Colour Training UK
Event registration page: https://www.eventsforce.net/bad/483/home
Description of activity: You are warmly invited to the Skin of Colour Training (SOCT) UK 2025. In case you haven’t heard of us before, we are a professional educational organisation in the UK made up of Dermatologists and Doctors with the primary aim of providing excellence in Skin of Colour education. We have run 3 consecutive training conferences aimed at healthcare professionals which have been very successful.
For the first time, we are delivering a half day pre-meeting aimed at professionals outside of healthcare (e.g. hairdressers, aestheticians and the general public) , who are interested in learning about dermatological concerns in skin of colour.
The meeting is part of Eucerin UK’s Social Mission programme aimed at reducing health inequalities in the U.K and we have also partnered with the British Skin Foundation to ensure that we reach the right audience.
Where? The Royal College of Physicians, London.
When: Wednesday 22nd of January 2025
Time commitment: Half-day
Campaign supporter: Eucerin UK’s Social Mission programme /British Skin Foundation


Please note:
Organisation or institution: The Dudley Group NHS Foundation Trust / University of Nottingham
Main study contact: [email protected]
Public contact details: [email protected]
Description of activity: This study aims to run focus groups that aim to understand patient's experience of living with anogenital psoriasis and discuss treatments they have used.
Recruit criteria: Any adult with an anogenital psoriasis diagnosis (by a dermatology specialist). Interested in participating in a research project looking at the treatment options of anogenital psoriasis. Able to attend an online focus group with 3-5 participants.
Where? Online via Microsoft Teams
Time commitment: 1 - 2 hours
Closing date: 01.03.2025 (may be extended)
Funder/sponsor: UK Dermatology Clinical Trials Network (UKDCTN)


Please note:
Organisation or institution: University of Aberdeen
Main study contact: Professor Gareth Jones
Public contact details: Email: [email protected] or phone: 01224 437562
Description of activity: The BSR-PsA register is a long-term observational study which aims to investigate the impact of psoriatic arthritis on patients’ quality of life and to monitor the effectiveness and safety of treatments. There are two main forms of data collection: (1) Clinical data collected from a patient’s medical notes; and (2) Questionnaires completed by study participants. Some patients may also be asked to donate some blood and urine samples for storage in a biobank.
Recruit criteria: People with a diagnosis of PsA who meet the CASPAR criteria
Where? UK hospitals who are a participating centre. Please ask your local rheumatology team if they are a recruiting site.
Time commitment: Annual questionnaire completion, and at 3 and 6 months if a patient is starting a new biologic therapy.
Closing date: December 2025, but recruitment may be extended.
Other information: If your local rheumatology service is a recruiting site, they will assess whether you are eligible for the study. Please see the Patient Information Sheet for more information.
Funder/sponsor: British Society for Rheumatology
Please note:
Organisation or institution: Intelligent Fieldwork
Main study contact: Nicola Watkins
Public contact details: [email protected]
Description of activity: Intelligent Fieldwork recruit people to take part in paid medical market research.
We have a study where the client, a medical device design company, is designing new injection devices which will hopefully be developed to make patient's lives easier.
They are looking to speak to women who have PSA and Psoriasis, and other autoimmune conditions which may affect strength, dexterity and fatigue. ALL medical devices have to go through rigorous testing by potential end users in order to come to market, so it really isn't just market research.
Sessions will last for up to 1-hour, in either Bristol or London. Participants will receive £100 by bank transfer or PayPal for their time and opinions. There are NO needles or medication involved, this is not a clinical trial. And certainly NO sales. It is simply to gather the opinions of potential end-users of the device.
We work within a lot of industry guidelines, such as BHBIA (British Healthcare Business Intelligence Association); MRS (Market Research Society) and obviously with the GDPR guidelines. People will be told exactly what is happening with their data.
Typically interviews are audio and video-recorded but will only be stored for the duration of the study and will only be viewed by people from the project team and for analysis purposes only. Once the findings of the study are reported, participants data is anonymised and therefore would not be traceable back to them.
We take patients’ information, data and well-being very seriously. Without their feedback products would not be nearly as good as they are and so we are enormously grateful that they will take the time to take part in these studies.
Recruit criteria: Female participants over the age of 12 years
Where? London or Bristol
Time commitment: 1 hour
Closing date: End of October 2024
Funder/sponsor: Crux Product Design


Please note:
Organisation or institution: University of Aberdeen
Main study contact: Claire Hunter
Public contact details: [email protected]
Description of activity: Some people with psoriatic arthritis have difficulty maintaining a healthy body weight, but we don’t know exactly why this is, or why this affects some people and not others.
We do know that there are lots of factors which can influence body weight and having a rheumatic disease can also affect these factors.
The aim of this study is to find out whether people with psoriatic arthritis who have excess body weight or low body weight have different lifestyle habits to those who have a heathy body weight.
We also aim to understand the impact of chronic pain, and reduced mobility and function on your lifestyle habits.
Whether you have lost or gained weight, or your weight hasn’t changed since your diagnosis, we are interested in hearing from you.
This will help us understand the factors that both help and hinder body weight maintenance. There are two phases of this study:
1) an anonymous online survey; and
2) an optional follow-up interview.
Recruit criteria: People living in the UK who have been diagnosed with psoriatic arthritis by a rheumatologist, are aged 18 and over, can speak English, and have never had an eating disorder (anorexia nervosa, bulimia nervosa, binge eating disorder, orthorexia, ARFID)
Where? Remote, online (survey) / video call (interview)
Time commitment: The online survey will take approximately ten minutes to complete, and the optional interview will last between 30 and 60 minutes.
Closing date: The survey closes on 8 August 2024. Interviews will take place during August and September 2024.
Other information: To learn more about this study, or take part, please read the participant information sheet:
Funder/sponsor: University of Aberdeen
Organisation or institution: Atlas Primary/Virtue Research
Contact: [email protected]
Description of activity: PsA patient's can share their experience on their treatment journey, by participating in a 15 minute survey that would compensate with a $35 check when the study closes out.
To apply, if qualified complete the survey: https://app.virtueresearch.com/c/LIGRBPNC
Recruit criteria: Psoriatic arthritis patient's in the USA, >18 years of age, currently receiving treatment.
Where? USA
Time commitment: For the next 30 days
Closing date: 21 June 2024
Funder/sponsor: Atlas Primary


Please note:

Organisation or institution: Atlas Primary
Contact: [email protected]
Description of activity: 7 waves of research are being conducted for psoriasis and the goal is to reach 100 patient's with each wave of research with a 15 minute long survey.
Recruit criteria: Psoriasis patient's in the USA, >18 years of age, currently receiving treatment.
Where? USA
Time commitment: Each wave is a month long.
Closing date: Each month, will have a new wave for patient's to participate in.
Funder/sponsor: Atlas Primary

Please note:

Organisation or institution: Amgen
Contact: [email protected]
Description of activity: The study will aim to estimate the efficacy of apremilast compared with placebo in the treatment of juvenile psoriatic arthritis (JPsA) in pediatric participants 5 to less than 18 years of age.
Recruit criteria: Male or Female participants 5 to < 18 years of age at the time of randomization. Participant must have a confirmed diagnosis of juvenile psoriatic arthritis (JPsA).
Where? Birmingham, Nottingham
Time commitment: Start dates may differ between countries and research sites.
Closing date: November 2027
Other information: Study is listed on https://bepartofresearch.nihr.ac.uk/
Funder/sponsor: Amgen
Please note:
Institution: Accellacare Clinical Research
Main study contact: Emilia Kudanowska
Public contact details: www.improvingtreatments.co.uk
Description of activity: Are you or anyone you know living with moderate to severe plaque psoriasis?
If so, consider participating in our psoriasis research study. You may be eligible for the study if you:
About this study:
Disclaimer: You may or may not benefit medically from taking part and there is absolutely no obligation to participate. If you are eligible and do decide to join the research study you are under no obligation to continue and may withdraw at any time and do not have to give us a reason for doing so.
Recruit criteria: Psoriasis patients.
Where? Corby, Northamptonshire/ Coventry, Warwickshire/ Shipley, Yorkshire/ Northwood, North London/ Orpington, South London
Time commitment: 64 weeks
Closing date: May 2025
Funder/sponsor: Leo Pharma
Please note:
We have a continual process of data gathering, which we do via all the processes of joining PAPAA. This largely unpublished data gives us a view of our demographic group, therefore allowing us to tailor our material to suit.
We also have been surveying people with psoriasis and psoriatic arthritis since our early origins for more than 20 years. Those rich qualitative data provides us with a real life view and also helps us to put forward a collective consensus of what it is really like to live with both psoriasis and psoriatic arthritis.
You can be part of that process and also know that whatever you say will be confidential and anonymous, it’s up to you.
We have now developed the PAPAA survey, which aims to build and add to those previous data results.
We will not ask for your name or contact details, all submission will therefore be anonymous. There are a few compulsory sections, to help us understand who we represent.
About you: The answers you give will help us to raise the true impact and how it affects real people, by providing a UK wide picture.
Quality of life: In order to give the true effect of psoriasis and or psoriatic arthritis, we would like to know how it affects your day-to-day life.
Research priorities: As someone who lives with a chronic disease, you have a unique insight into the issues that are important to you. We would like to gather these issues together and create a list of priorities that are important to people with psoriasis and psoriatic arthritis.
Final comments: Your chance to say anything else not covered in the other sections, these comments will help us to provide rich information that has context and is meaningful.
Be part of the bigger picture and get your opinions and views logged, so we can tell those who need to know exactly what you and others with these conditions actually feel, think and want to be said.
Data from the PAPAA survey has been used in submissions to support the patient perspective in health technology assessments, with the National Institute for Health and Care Excellence (NICE), the Scottish Medicines Consortium (SMC) and the All Wales Medicines Strategy Group (AWMSG).
Institution: University of Oxford
Main study contact: Professor Laura Coates
Public contact details: [email protected]
Description of activity: We are planning to launch a European online study in June of this year to monitor people with psoriasis to see who develops PsA. The study will be completely remote with participants filling in questionnaires online and sending small fingerprick blood samples by post.
We are aiming to recruit 25,000 people with psoriasis across 10 countries. We think this cohort of patients will help us to completely change how we think about the development of PsA and how to predict this.
It will help us to design prevention studies, where we would offer potential drug treatments or lifestyle interventions (like exercise, stress management etc) to see if we can prevent PsA. We have really carefully designed the study with a number of patient partners across Europe.
Recruit criteria: People with psoriasis but not (yet) PsA
Where? All remote
Time commitment: Approx 20 mins every 6 months
Closing date: June 2024
Funder/sponsor: IMI funded consortium
Please note:
We would like to understand what is important to people with psoriasis and or psoriatic arthritis, when using internet search engines for answers to questions.
By completing the survey you will be helping us to endeavour to meet your needs.
This is your opportunity to let others know what it is like to live with these conditions.
How you were diagnosed, the treatments you received, how the appointment was delivered, or anything else that you want to share that you feel would help others in their journey living with psoriasis or psoriatic arthritis.
It's up to you, if you just want to get something off your chest, here is your chance.
You can submit anonymously or share your name.
We will add your personal journey to this website and with your help build a picture of the extent these conditions have on real people and their lives.
Please note: We will not publish your name and may edit your submission to protect your privacy and that of others mentioned.
Activity title:
Organisation or institution: Choice Health
Main study contact: Pauric Greenan
Public contact details: [email protected]
Description of activity: We work in clinical trials and research (specifically in the field of imaging) and we currently have a paediatric psoriatic arthritis study that requires a review of the language used in the Patient/Participant Information Sheet (Informed Consent Form). We would require 3 reviewers who generally will have experienced the condition previously (and/or a parent). We need comments on the docs shared with you and also to complete the review questionnaire attached.
Recruit criteria: 3 Past patients/parents of patients
Where? remote
Time commitment: 1-3 hrs each
Closing date 8th January 2024
Other information: Any questions, please contact Pauric on email above or 07880006046
Funder/sponsor: Choice Health
Organisation or institution: University of Oxford.
Main study contact: Lija James
Public contact details
Email: [email protected]
or through QR code registration
Description of activity: We are looking for people to join our patient partner group for the COLIPSO study. This study will investigate the extent of liver disease in psoriatic disease. Patients with psoriatic disease have a much higher risk of developing non-alcoholic liver disease.
This causes significant impact on their life and is a difficult clinical problem as some therapies for psoriasis and arthritis may worsen the liver disease. Previously the only tests available have been blood tests (which are often normal until the liver is quite damaged) or a liver biopsy (an invasive and risky procedure which only takes a very small sample of the liver).
Our colleagues have developed and tested a new type of MRI scan which can measure inflammation and scarring in the liver allowing us to study liver disease in detail.
We want to use this new technology to study the proportion of people with psoriasis who also have associated liver disease and measure how severe this is. We will use blood and stool samples to study why and how the liver disease develops in many people with psoriatic disease.
We will repeat the scans and research samples after 6 months on a new treatment to see how common psoriasis treatments affect both inflammation and scarring in liver disease.
Recruit criteria: Individuals with a diagnosis of psoriatic arthritis.
Where? Remote session on Zoom
Time commitment: 1 hour online session
Closing date: 19/12/2023
Funder/sponsor: National Psoriasis Foundation. Perspectum. UCB Biopharma SRL.
Please note:
PREDICT - Psoriatic Arthritis Recognising Early Disease Indicators Conferring Treatment response
University of Oxford
Dylan McGagh
Email: [email protected] or through QR code registration
We are looking for people to join our patient partner group for the PREDICT study. This study will investigate whether blood tests can predict which patients will respond to different treatments. This study will also test the use of digital tools such as smartwatches and apps to capture more about how patients with psoriatic arthritis feel and function in response to different treatments. We want to gather the perspectives of individuals with psoriatic arthritis on our overall study aims, gain some feedback on our study specific app and more broadly ask on perspectives of using digital measures to capture information on symptoms and mobility.

Individuals with a diagnosis of psoriatic arthritis
Remote session on Teams / Zoom
1.5 hour online session
10/12/2023
British Psoriatic Arthritis Consortium (Brit-PACT)
Please note:
The Living with a LTC Study
King's College London
Dr Annie Jones
We know that psychological distress (anxiety and depression) is common in and experienced differently by people living with long-term health conditions, such as psoriasis. Being able to better measure and understand how people living with a LTC experience psychological distress would allow us to provide interventions specifically tailored to the challenges of living with a LTC and therefore provide the most appropriate support for these people. We have developed a questionnaire and would like to test it. Taking part involves completing 2 short online surveys (10 minutes; 5 minutes), a week apart. Participants have the option of being entered into a prize draw to win 1 of 5 £100 Amazon vouchers.


People >18 in the UK who have a diagnosis of a physical long-term condition (e.g., psoriasis, psoriatic arthritis and others)
Remote
15-25 minutes
31/01/2024
Please contact [email protected]; [email protected] and [email protected] with any questions.
King's College London
Please note:
Institution: University of Liverpool
Main study contact: Megan Devlin
Public contact details: [email protected]
Description of activity: My name is Megan Devlin, and I'm a third year psychology undergraduate student at the University of Liverpool. For my final year dissertation, I'm conducting a research study (with my supervisor) exploring how living with a chronic physical illness can impact romantic relationships. I'm really passionate about psoriatic arthritis (PsA) and how it can impact relationships because my mum was recently diagnosed with it, and I can see first-hand how difficult it can be to adjust to a new normal. The study will involve a short interview, lasting roughly an hour, and involve questions about how life was like before and after your diagnosis, how your relationship may have changed and your thoughts and feelings about this.
Recruit criteria:To take part you must: be aged 18 or over, be in a romantic relationship, have a formal diagnosis of psoriatic arthritis, speak fluent English, and be happy to talk about personal experiences.
Where? The interviews can take place virtually (via Zoom or Microsoft Teams) or in-person on the University of Liverpool campus, depending on your personal preference.
Time commitment: The interview will last approximately 1 hour.
Closing date: early December 2023
Funder/sponsor: University of Liverpool
Please note:


Institution: Wisdo Health - a social health app
Main study contact: Boaz Gaon, Wisdo CEO
Public contact details: [email protected]
Description of activity: Wisdo Health are a well known social support and health app currently partnered with UCB, the global biopharma company.
We are looking for up to 20 UK based individuals who'd be interested in joining a paid pilot, during which they will help us identify how to improve our digital community offering - with the goal of reducing loneliness and social isolation amongst people living with Psoriasis, and potentially other dermatology conditions.
Participation is very low effort and includes:
1. Registering to the Wisdo app, from the comfort of your home.
2. Engaging at your leisure with the app for 60 days, including chatting once a week 1:1 and 1: group with other members.
3. Sharing ongoing measurements, feedback and insights with our project manager. We pride ourselves on being patient centric, so we are very much trying to make sure that we onboard individuals who can be generous with their insights and time.
Wisdo Health will compensate members for their time and generosity. Thank you!
Recruit criteria: UK based adults with psoriasis or HS (hidradenitis suppurativa).
Where? Remote. From the comfort of your home with flexible hours.
Time commitment: Approx 20 mins every 6 months
Closing date: October 1 2023
Funder/sponsor: Wisdo Health
Please note:


A London based casting agency specialising in finding real people, with real stories, for commercials and documentaries around the world is working on a new campaign for a skincare brand.
For this campaign they are looking for 'Skin Heroes' real people with challenging psoriasis, eczema, dermatitis etc. The tone of the film is joyous, warm and unapologetic, and they are specifically looking for people for which their skin condition doesn’t hold them back from enjoying life and having a pep in their step.
They are particularly looking for the following people:
But open to anyone who wants to apply

Fee: This is a paid opportunity (exact amount TBD).
Dates: Filming will take place on either 30th or 31st August.
Location: People can be based anywhere in the UK.
If you are interested email: [email protected]
Closing date to apply 8th August 2023
Please note:
Organisation or institution: iPROLEPSIS https://www.iprolepsis.eu
Main study contact: Laura Coates
Public contact details: https://www.iprolepsis.eu/contacts
Description of activity: We are looking for people with psoriatic arthritis to help with a survey. This survey is part of the iPROLEPSIS project, funded by the European Union. With a duration of 4 years this project aims to develop a digital health system for the care of psoriatic arthritis patients.
Within iPROLEPSIS we want to apply technology, such as smartphones and smartwatches to help you monitor your disease and help you find the coping mechanisms that suit you.
We are at the beginning of the development process and would love your input on various ideas. Your input will be used to create user requirements for the developed services. So your opinion counts!
All data is confidential and completely anonymous. It will take about 20-30 minutes to fill out this questionnaire. Your efforts will be well appreciated.
Recruit criteria: People diagnosed with psoriatic arthritis
Where? Online survey
Time commitment: 20 - 30 minutes
Closing date: 31 July 2023
Funder: European Union
Please note:
Organisation or institution: University of Oxford.
Main study contact: Caroline Struthers.
Public contact details: Link to form
Description of activity: We are looking for people to help with a new project called Trialblazers. We are aiming to improve the way trials are designed and carried out by involving you from the very beginning.
We are holding an in person "Build-a-Trial" workshop for people with mild or suspected psoriatic arthritis in Oxford on: 18 July from 10.30-2.30pm.
We will also be holding two two-hour online Zoom workshops on 27 July.
During the workshops, we will ask for your input into key decisions about a new trial testing the best ways to treat mild psoriatic arthritis.
These decisions could include how to recruit participants, acceptability of treatment alternatives, which symptoms are most important to treat, and how to measure the treatment effects in the best way.
Recruit criteria: You will have mild (or suspected) psoriatic arthritis, or be a friend or family member; over 18.
Where? In-person workshop, Exeter Hall, Oxford; Online workshops will be on Zoom.
Time commitment: In-person workshop will be from 10.30am-2.30pm. No preparation needed. Online workshop will be from 2-4pm and 5-7pm.
Closing date: 23 July 2023.
Other information: Workshop participants will be paid £100 plus reasonable expenses (travel, childcare etc.).
Online participants will be paid £60 per workshop plus appropriate refreshments supplied by post!
Funder/sponsor: University of Oxford, Participatory Research Fund.
Please note:
Organisation or institution: Just-Worldwide and Branding Science
Main study contact: Kristen Saylor
Public contact details: Kristen@just-worldwide, 609-955-8607
Description of activity: This is a year long research study regarding the GPP Patient experience. It consists of 4 phases ( roughly one for each quarter of the year) Each phase will include: - 10min intro call (just one quick call during project life cycle) - 10min online community task - 60min WATDI call - 15min online/ppt pre-task - Flare diary is an optional component Qualified respondents that participate in this project will be paid for each assignment completed as well as a bonus when all tasks are completed in a each phase
Recruit criteria:
Where? Remote
Time commitment: 1 full year
Closing date: 17 March 2024
Funder/sponsor: Branding Science LLC.
Please note:
Organisation or institution: A student from Walden University PhD program.
Main study contact: Katie McCann
Public contact details: [email protected]
Description of activity: The participant will be interviewed with about 13 questions on their experiences living with psoriatic arthritis and their quality of life.
Recruit criteria: men and women between the ages of 18-25. Primary diagnosis of PsA. Living in the United States.
Where? Remote, in-person (Nashua, NH), phone call, or Survey Monkey.
Time commitment: 60-90 minutes.
Closing date: 31 August 2023
Funder/sponsor: Katie McCann
Please note:
Organisation or institution
Department of Rheumatology and Clinical Immunology at the Amsterdam UMC, location AMC
Main study contact: Dr. Marleen van de Sande
Public contact details: [email protected]
Description of activity: Up to 1 in 3 people who have psoriasis can go on to develop a related arthritis causing joint pain and disability.
Researchers are exploring whether it might be possible to treat people who have psoriasis to reduce their risk of developing psoriatic arthritis.
This survey aims to understand what medications or lifestyle changes would be acceptable to people with psoriasis to reduce their chances of developing psoriatic arthritis in the future.
Understanding patients’ preferences is important to ensure future treatments of this kind are efficient and acceptable to patients. This survey is a one-time questionnaire that should not take longer than 20 minutes to complete. Participation is voluntary, and all responses to the survey will remain confidential and anonymous.
This study is in collaboration with Dr. Laura Coates (Principal investigator, Associate Professor, rheumatologist Oxford), Dr. Marie Falahee (Lecturer in Behavioral Rheumatology, University of Birmingham), Dr. Jorien Veldwijk (Assistant-professor, Erasmus School of Health Policy & Management), Prof. Stephen Pennington (Professor Of Proteomics, University College Dublin) and Prof. Oliver FitzGerald (Newman Clinical Research Professor, University College Dublin).
Recruit criteria: You can take part in this survey if you:
Where? Remote, it's an online survey
Time commitment: 20 minutes
Please note:
Organisation or institution: Global Patients
Main study contact: Miryam
Public contact details: [email protected]
Description of activity: Task and interview
"At Global Patients, an IQVIA business, we are looking for people with psoriasis from UK to participate in a 60-minute interview and to do a previous 60-minute task.
We will compensate the participants with £120 as a token of appreciation for their time. There is no medical treatment being provided as part of this study. All data collected during this research will be treated anonymously, confidentially and analysed in aggregate form.
If you are interested in participating or someone you know, please do not hesitate to contact [email protected]"
Recruit criteria: Psoriasis patients
Where? In the UK
Time commitment: 1-hour interview plus a 60 min pre-task
Closing date: End of February 2023
Funder/sponsor: Global Perspectives
Please note:
Organisation or institution: Channel 4
Main study contact: Codie Lu Scott
Public contact details: [email protected]
Description of activity: Could you provide a loving home for a rescue dog? The Channel 4 series, The Dog House is looking for couples, individuals, friends and families who are seriously considering bringing a rescue dog into their lives and are willing to share their reasons for doing so.


Recruit criteria: People who would like to adopt a dog and are open to sharing their reason why. We are particularly looking to share how much a difference a dog can make to people’s mental health whilst living with a skin condition. We understand that with a skin condition come some serious anxieties. Studies have shown that even small interactions with dogs cause the human brain to produce oxytocin, a hormone often referred to as the “cuddle chemical.” Oxytocin increases feelings of relaxation, trust, and empathy while reducing stress and anxiety. Where?
Location: Woodgreen Pets Charity
Time commitment: 2 days approx.
Closing date: 23/03/23
Activity title: Research on atopic dermatitis
Organisation or institution: Exafield - market research
Main study contact: Laurie Chardon
Public contact details: [email protected]
Description of activity: We are currently conducting a market research study on atopic dermatitis (AD), and we would like to speak with you to explore some aspects of living with the disease.
We are looking for patients. It will be an online research where we will ask the patient to download and test a new app on their smartphone.
The patient will be reimbursed £10 to download the app, complete the little questionnaire and confirm us (10min maximum in total).
ONE Inclusion criteria: - Been formally diagnosed with atopic dermatitis
Recruit criteria: Atopic dermatitis
Where?: Remote
Time commitment: 10 min
Funder/sponsor: We Fight
This is an external to PAPAA opportunity. If you are interested, please contact the organisers direct.
Can you help a team of researchers?
A group of rheumatologists, dermatologists and people with joint disease and other medical professionals are working together to improve care for people with psoriatic arthritis, through the British Psoriatic Arthritis Consortium (BritPACT).
There are many highly effective medications (biologics) available in the UK to treat psoriatic arthritis but access to them can vary around the country.
The team are seeking to understand people’s experience of accessing biologic medicines (good or bad) and how effective they have found them.
Your input will help the development of future research projects and understand questions that are important from the perspective of people living with psoriatic arthritis.
Your input and time taken to complete this survey is valued highly. Your responses will all be anonymous.
Have you ever wondered whether what you eat affects your psoriasis? This is one of the commonest questions that people with psoriasis have. At present, there is not enough high-quality research to be able to be able to make specific recommendations. To help answer this question, we have launched The APPLE Study!
This is the first study of its kind in the UK. We aim to find out if there are certain diet and lifestyle patterns that are more commonly found in people with more severe psoriasis compared with those with milder forms. This research will help us understand if dietary changes could be helpful for psoriasis management in the longer term.
Participation in The APPLE Study will involve the completion of:
The APPLE Study will take place online. You will need internet connection to access the online survey and diet diary.
We strongly encourage you to complete The APPLE Study on a laptop, or desktop computer as some of the questions will not display properly on a smartphone.
On completion of The APPLE Study you will receive:
Eligibility requirements
Email: [email protected].
Instagram: the_apple_study
Exploring the role of diet in the management of psoriasis: Dietary behaviours, perceptions and self-reported skin outcomes of people with psoriasis
The University of Hertfordshire, Nutrition and Dietetics Department
Poppy Hawkins: [email protected]
Email - Poppy Hawkins: [email protected]
We are investigating dietary habits in people with psoriasis and how this impacts their symptoms and long-term management of their condition. We also want to understand more about which diets people with psoriasis are trying, and where they go for information, and experiences. Little research has been done in this area. By taking part in this study, you will be helping us to understand how best to support people with psoriasis, in the absence of clear dietary guidance. This is an anonymous online questionnaire which will take about 25 minutes to complete. This study has been approved by The University of Hertfordshire Health, Science, Engineering and Technology Ethics Committee with Delegated Authority, reference: aLMS/PGR/UH/05068(1).
People who have been diagnosed with psoriasis, over the age of 18 years old, can read, write and speak English fluently and live at a UK address (but not in an institution)
Remote - Online questionnaire. There is an option to do it over the phone if someone is unable to do it online. Just contact Poppy Hawkins ([email protected]).
An estimated 25 mins
Other researchers involved. Supervisor, Dr Rosalind Fallaize ([email protected]), Dr Kate Earl ([email protected]) and Dr Athanasios Tektonidis ([email protected]); each of which have extensive experience in large nutritional studies, epidemiology, and clinical nutrition. Those who complete the questionnaire will be given the option to participate in a further diet and psoriasis study, if they wish to.
This is being done as part of a QR-funded PhD Studentship, titled ‘The role of diet in the management of psoriasis’ 2021-2024. At the University of Hertfordshire.
Activity title: Comparison of Wynzora Cream and Enstilar Foam in the Treatment of Psoriasis - a Study on Patient Preference.
Organisation or institution: The Center for Dermatology and Dermatologic Surgery.
Main study contact: Dr. Bev Fontaine
Public contact details: text 202-695-0409 or Call 202-955-5757
Description of activity: The purpose of this study is to investigate whether people with psoriasis prefer applying a cream formulation or a foam formulation of study medication to their skin. we are comparing two medications used for the treatment of psoriasis. Their only difference is that one is foam and the other is a cream. Participants in this study will have both products applied to their skin. They will then complete a series of questionnaires to determine which product they prefer.
Recruit criteria: 18 years of age or older; No history of hypercalcemia; high vitamin D; or significant renal or liver disease.
Where?: The study visit will take place at The Center for Dermatology at 2311 M street, Washington DC 20037.
Time commitment: There is only one visit needed. The visit will last approximately 2 hours.
Other information: Qualified participants who complete the study will receive a stipend payment for their time. Participants may also receive study-related medication and care for the duration of the study.
Funder/sponsor: Research is carried out at the Center for Dermatology and is sponsored and financed by Almirall. It has reviewed by an IRB.
Please note: PAPAA has no connection with this study or the sponsors and has not received any funds or payment of any kind, for highlighting this opportunity.
People with mild to moderate plaque-type psoriasis are needed for a research study.
You must be aged 18 to 65 years old and have a confirmed diagnosis for at least 6 months.
Smokers of up to 20 cigarettes, or equivalent per day allowed. It involves 1 session of 4 nights’ residence and 9 outpatient visits, over 3 months.
Study fee: £2,950 plus reasonable travel expenses.
You might not be suitable, if you’re taking certain medications
If interested, you will need to contact HMR directly
Email: [email protected]
Telephone: 0800 783 8792
Please note: This activity is independent of PAPAA.
A 4th Year Medical Student at Cardiff University, and with his supervisor is currently working on a project titled Medical Photographs: Patient perceptions of medical photography of dermatological conditions. This project has obtained ethical approval from Cardiff University School Research Ethics Committee.
Dermatology is a visual specialty, and images of dermatological conditions are important in the education of future clinicians in the specialty. The majority of images of dermatological conditions in major UK textbooks are of Caucasian patients resulting in an incomplete picture of dermatological conditions in various skin tones, as brought to the attention of many by a recent petition.
This pilot study is an attempt to understand some of the patient factors behind the reasons for inadequate representation of patients of skin of colour in dermatology images. In addition, they would also like to determine if there are any other factors that influence a patient’s consent to medical photographs of their dermatological conditions. Therefore, the aim of the study is to determine if patient demographics influence perception of medical photography of dermatological conditions.
The study contains a short questionnaire made up of 4 headings, including questions asking participants: information regarding their sex, age, ethnicity, information relating to socio-economic status, and religion; and their opinions on medical images being taken of their dermatological conditions in different scenarios.
These questions will be asked to find out if these different factors influence patients’ perceptions on consenting for medical photographs being taken of their dermatological conditions, or if it influences whether health care professionals will request consent from patients.
The questionnaire will take approximately 5-10 minutes to complete.
This opportunity is now closed
Updated inclusion criteria for weight and psoriasis research.
A project is being undertaken at King’s College London called: Feelings, Emotions And Behaviours In Weight And Psoriasis. People living with psoriasis often describe weight management as a challenge. Several factors are likely to explain why managing weight alongside psoriasis can be difficult.
The inclusion criteria has now changed for this research. Ethical approval has been obtained to include people with a Body Mass Index (BMI) of below 25 (<25 BMI) as well as the previous above 25 (>25 BMI).
These may include psychological factors such as beliefs about weight and psoriasis, eating and physical activity behaviours, and how one feels in their skin. Thus, traditional weight loss interventions that focus on diet and physical activity are unlikely to be relevant to the challenges people with psoriasis face.
The purpose of the project is to look beyond the role of diet and physical activity for weight management in psoriasis. It will aim to find out what other factors such as feeling, and emotions can present as a challenge when people with psoriasis want to lose weight. Building a holistic understanding of weight management in psoriasis is a priority to ensure patients’ needs are addressed sufficiently and adequately.
This can support the design of a more tailored weight loss approach which ultimately can lead to improved patient outcomes.
To learn more and to read the information sheet about getting involved, follow this link.
This study is now closed.
Sheffield Hallam University
Dr Elaine Clarke
We would like people to take part in a one-off online survey at a time that is convenient to them. The survey questions will ask for some demographic information, information about the person's psoriasis and its treatment(s), and about how they normally respond to difficult situations. We expect taking part in the survey to take around 15 minutes.
People who have been prescribed topical treatments for psoriasis that have an active ingredient (i.e., not just moisturisers/emollients)
Remote
Approx 15 minutes
The survey is now closed
Sheffield Hallam University's Research Ethics Committee has approved this study.
